  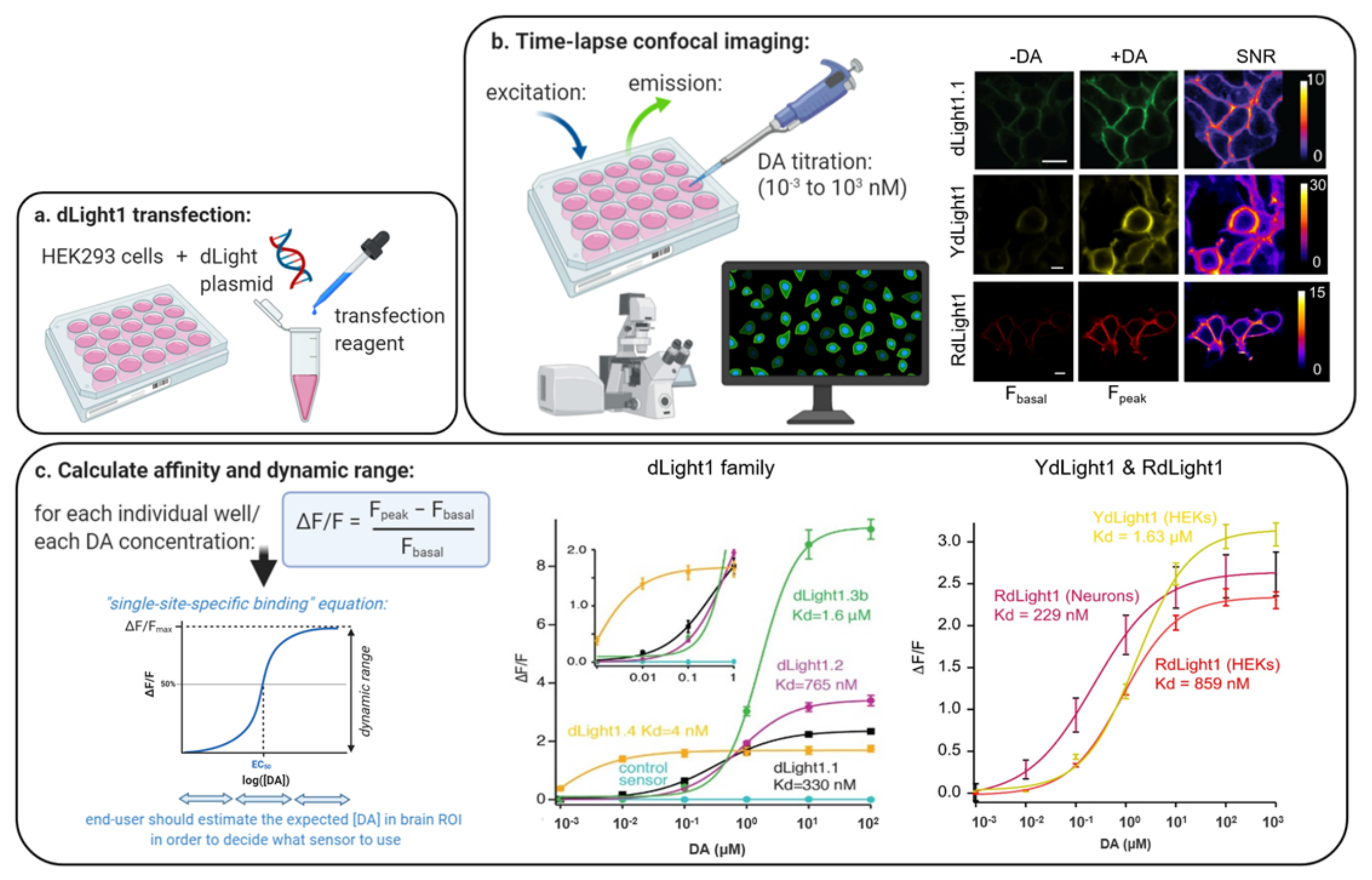
Several studies have already demonstrated the powerful application of viral expression of such sensors for studying DA dynamics in live animals and in brain tissue circuits using e.g., fiber photometry and microscopy techniques 6, 7, 8, 9, 10, 11, 12. These sensors allow the interrogation of extracellular DA levels with unprecedented spatiotemporal resolution using optical measurements of fluorescent intensity 6, 7. Important advances in the field of DA detection have recently been achieved with the development of G protein-coupled receptor (GPCR)-based sensors that directly couple the presence of DA with an increase in fluorescent. Sensitive methods to study DA neurotransmission in cell cultures, tissue preparations, and living organisms are necessary to gain mechanistic insights into DA signaling in health and disease states, and for the development of effective therapeutics that target dopaminergic circuits. In addition, the mechanisms through which DA exerts its short- and long-term effects on emotional states and behavior remain unclear. Still, we only have a limited understanding of the nature and progression of DA dysfunction in diseased states. The dopaminergic circuit of the brain is also a major target for several therapeutics used in the treatment of these movement and mental disorders, and for drugs of abuse 2, 3, 4, 5. In line with these functions, decades of research have implicated disturbances in dopaminergic neurotransmission in both movement disorders and mental illnesses 1. Importantly, the sniffer cell framework can readily be applied to the growing list of genetically encoded fluorescent neurotransmitter sensors.ĭopamine (DA) serves as a neuromodulator in the brain where it is critically involved in locomotor control and higher brain functions such as motivation and reward-related learning. Furthermore, we use the sniffer cells to measure dopamine uptake and release via the dopamine transporter as a radiotracer free, high-throughput alternative to electrochemical- and radiotracer-based assays. In proof-of-principle experiments, we apply the sniffer cells to record endogenous dopamine release from cultured neurons and striatal slices, and for determining tissue dopamine content. We generated sniffer cell lines with inducible expression of seven different dopamine sensors and perform a head-to-head comparison of sensor properties to guide users in sensor selection. Here, we present a blueprint for making dopamine sniffer cells for multimodal dopamine detection. However, these sensors’ utility for in vitro and ex vivo assays remains unexplored. Genetically encoded fluorescent dopamine sensors have recently enabled unprecedented monitoring of dopamine dynamics in vivo. Detailed data on dopamine dynamics is needed to understand how dopamine signals translate into cellular and behavioral responses, and to uncover pathological disturbances in dopamine-related diseases. Consistently, dopaminergic dysfunction is involved in a spectrum of neurological and neuropsychiatric diseases. Dopamine supports locomotor control and higher brain functions such as motivation and learning. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
